|
11/18/2023 0 Comments Change in entropy 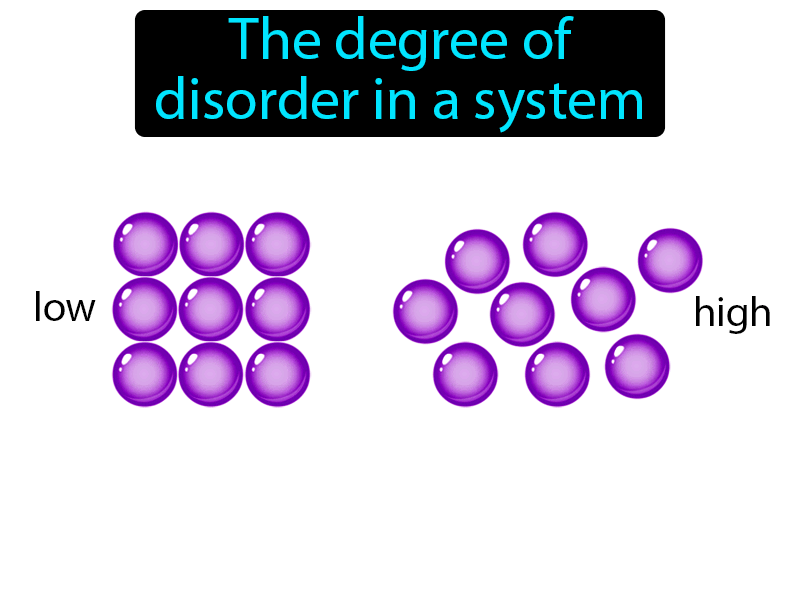 
(I say dimensionless, because it is measured using logarithms. Entropy Change can be defined as the change in the state of disorder of a thermodynamic system that is associated with the conversion of heat or enthalpy into. The proteins can't be readily undenatured, even if you change the conditions to thermodynamically favor denaturation because, kinetically, the proteins can't find their way back to their undenatured state.Īnyways, let's suppose we have a "reversible reaction", $$\ce = 11$, the opposite would happen: decreased entropy of the universe in the forward direction, increased entropy of the universe in the reverse direction (again, until it reached equilibrium). So, a change in entropy represents an increase or decrease of information content or disorder. 
By contrast, an example of an irreversible reaction would be cooking an egg, which causes the proteins to denature. Entropy changes accompanying a particular chemical reaction iviAi 0 are handled by writing (3.9. I.e., one where you can adjust the relative concentration of products and reactants back and forth as you change conditions. Noting that the number of microstates is labeled W W in Table 15.4 for the 100-coin toss, we can use S S f S i k ln W f-k ln W i S S f S i k ln W f-k ln W i to calculate the change in entropy. A reversible reaction is one that is kinetically reversible. Reaction B 2 H 2 (g) + O 2 (g) 2 H 2 O (g) There are 3 moles on the reactant side and only 2 on the product side. First, a defintion of terms: The meaning of reversible in "reversible reaction" is different from the thermodynamic meaning of reversible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
